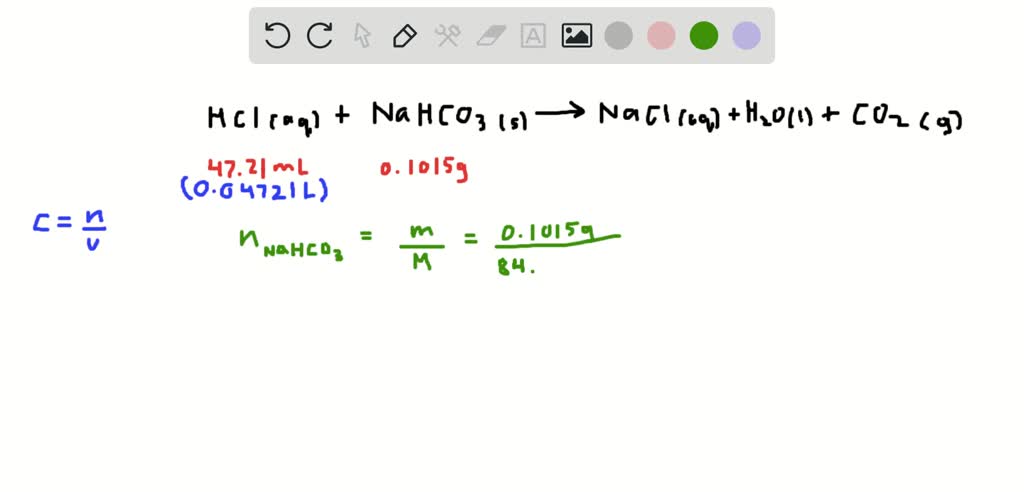
It is an ionic compound made of sodium cations (Na +) and bicarbonate anions (HCO 3- ). The sodium carbonate crystals obtained by doing so contain 10 molecules of water crystallization. Formula and structure: The chemical formula of sodium hydrogen carbonate is NaHCO 3, and its molar mass is 84.007 g/mol. CHO3.Na Net Charge, 0 Average Mass, 84.00660 Monoisotopic Mass, 83.98234 InChI, InChI1S/CH2O3.Na/c2-1(3)4 /h(H2,2,3,4) /q. The anhydrous sodium carbonate is dissolved in the water and recrystallized to obtain the sodium carbonate crystals. The Carbon dioxide so obtained in the below reaction is reusable. The obtained product that is sodium bicarbonate is then heated to get sodium carbonate which is our desired compound. 
Hence, it is removed by the process of filtration. Sodium hydrogen carbonate, ACS, 99.7-100.3, Thermo Scientific CAS, 144-55-8 Molecular Formula, CHNaO3 Molecular Weight (g/mol), 84.006 MDL Number. This addition to the NH 4 + and HCO 3 - leads to the formation of Sodium hydrogen carbonate which is not at all soluble. The reactions taking place can be represented as,Īfter that, add some amount of sodium chloride or common salt to the solution. Calcium chloride is an important byproduct in the process. The Ingredients involve, Sodium chloride or Salt Brine (NaCl), ammonia (NH 3), and Calcium carbonate or limestone (CaCO 3). Sodium hydrogen carbonate is used as a laboratory chemical, a leavening agent, a neutralizer, as an active component in fire extinguishers, as a source of. Sodium hydrogen carbonate is an approved food additive, E500, and is used. The ingredients involved in the extraction of sodium carbonate by the Solvay process are cheap and easily accessible. Sodium hydrogen carbonate - liberate carbon dioxide on gentle heating (or with acids). It is then precipitation out completely and is filtered and then finally ignited to get the sodium carbonate. By this reaction, the formation of sodium hydrogen carbonate takes place which is very slightly soluble is the presence of sodium ions. The molar mass or the molecular weight of sodium hydrogen carbonate is approximately equal to \(84.007\, \right)\) ions.Sodium carbonate is generally extracted through the following four processes:Īmong all the four the most common and the most used process is the Solvay process.ĭuring the process, carbon dioxide and ammonia are together passed through a cold saturated solution of sodium chloride. In sodium hydrogen carbonate, the oxidation state of sodium is \( + 1.\) In sodium hydrogen carbonate, the bond formed between the positively charged sodium ion and the negatively charged oxygen is an ionic bond. sodium bicarbonate (NaHCO3), also called baking soda, bicarbonate of soda, or sodium hydrogen carbonate, white crystalline or powdery solid that is a source. Who can purchase chemicals from us Findel Education will only sell to Academic Customers and will deliver only to the school address for.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
